
Je suis une CRO
ClinInfo
Who are we?

Je suis une CRO
CLININFO
ClinInfo SAS is a European company specialized in digital eClinical solutions for interventional or non-interventional clinical research.
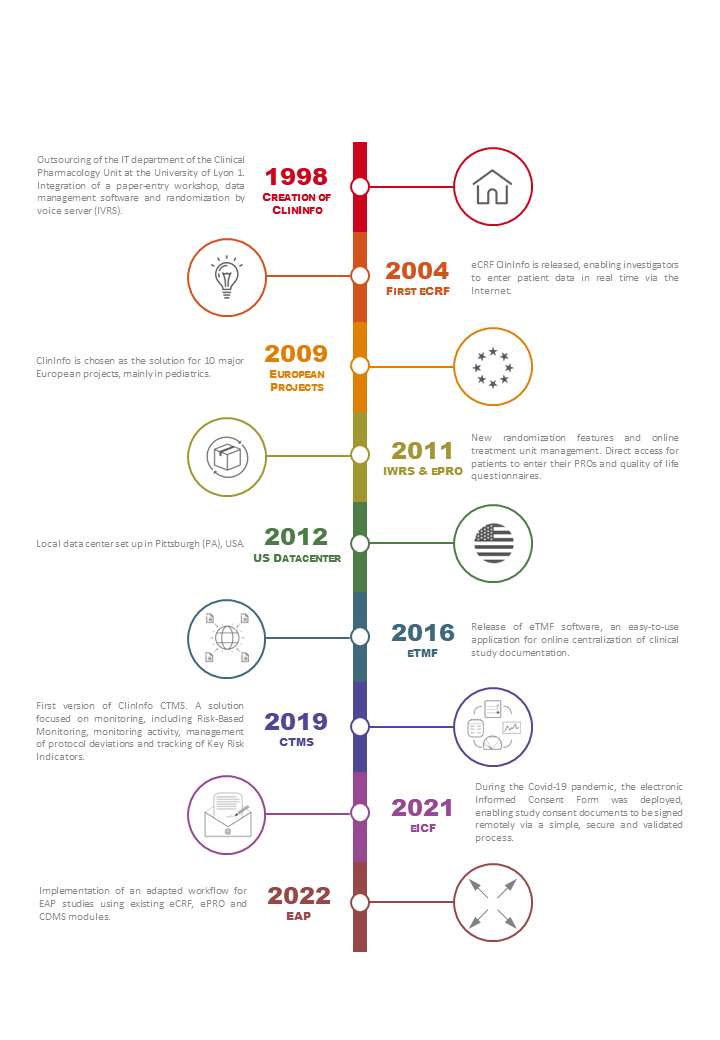
It was created in 1998 and comes from the Clinical Pharmacology Service of Université Lyon 1 (France), lead by Pr. Jean-Pierre Boissel.
With more than 23 years of expertise, we offer adapted and custom-made digital solutions, for BioTech experimental studies to large international clinical trials, in more than 50 countries.


Experiences
Since its creation 23 years ago, the Clininfo system has been used to manage clinical trials in a variety of fields such as cardiology, oncology, rheumatology, diabetology or pediatrics. Clininfo is involved in many international studies, and the system has been used by investigators and patients around the world. ClinInfo is also involved in seven European FP7 projects, as a partner and provider, and regularly works with the University College of Cork and the Paris Public Hospital Assistance.
In France, Clininfo is also a historical partner of Hospices Civils de Lyon (HCL) and provided its services for a large number of other structures, including most French universities and research centers.
In 2012, to promote our products and services to internationally, we established a data center in the United States, in Pittsburgh PA.




Missions
We create digital solutions to provide researchers in the health field with tools to carry out their clinical studies and thus improve their real productivity. Our approach coming from the academic and CRO domains, allows to really identify the needs and to accompany the customer to allow them to have reliable results.
This adaptability and flexibility are reflected by the quality of our products and the experience of our team, as well as the growing number of projects managed each year.
Our CSR Engagement
ClinInfo follows a responsible and respectful approach, in regards of its ecosystem
Ecology and sustainable development:
• Electricity supply from a local green energy provider (Urban Solar)
• Hosting provider with a strong ecological engagement (Avenir Télématique)
• Elimination of non-mandatory plastics (water bottles, cups, stirrers…)
• Donation of used office furniture for it to have a second life (chairs, shelves…)
• Purchase of new furniture made from recycled materials
• Elimination of paper documents to only keep electronic documents
• Waste separation
Social and societal:
• Gender parity among the team and strict equal pay based on experience
• Annual satisfaction survey of the team, four-monthly individual interview
• Staff awareness and prevention of discrimination (origin, religion, gender, disability…)
• Personal and professional life balance: flexible and adapted work hours, facilitated day off taking, generalized or occasional home office
• Collaboration with occupational medicine to prevent psychosocial risks
• Support of public research through an adapted economic model: cost sharing, budget limitation to what is mandatory
• Transparency culture towards our clients: explicit costs, clear and unambiguous contracts, proof documents provided

