
Je suis une CRO
CDMS , conduct your project
using a powerful oversight tool.

Je suis une CRO
CDMS
ClinInfo's CDMS is used to conduct your project through its lifecycle.
Using a set of oversight and coordination tools, you can manage various aspects related to the solution setup, collected data or user profiles.
Our CDMS shows in real-time the progress status of your project, making the follow-up easy for data managers, project managers, sponsors and statisticians.
Patient's data
ClinInfo's CDMS offers a wide range of functions to view, control, lock or export data. All actions are traced into a fully automated audit trail that can be consulted at any time, with a real transparency of actions performed.
Our CDMS also includes numerous tools such as real-time reports, progress charts, missing data reports or database quality metrics. More than 50 reports are included in the default application, and others can be created on demand.
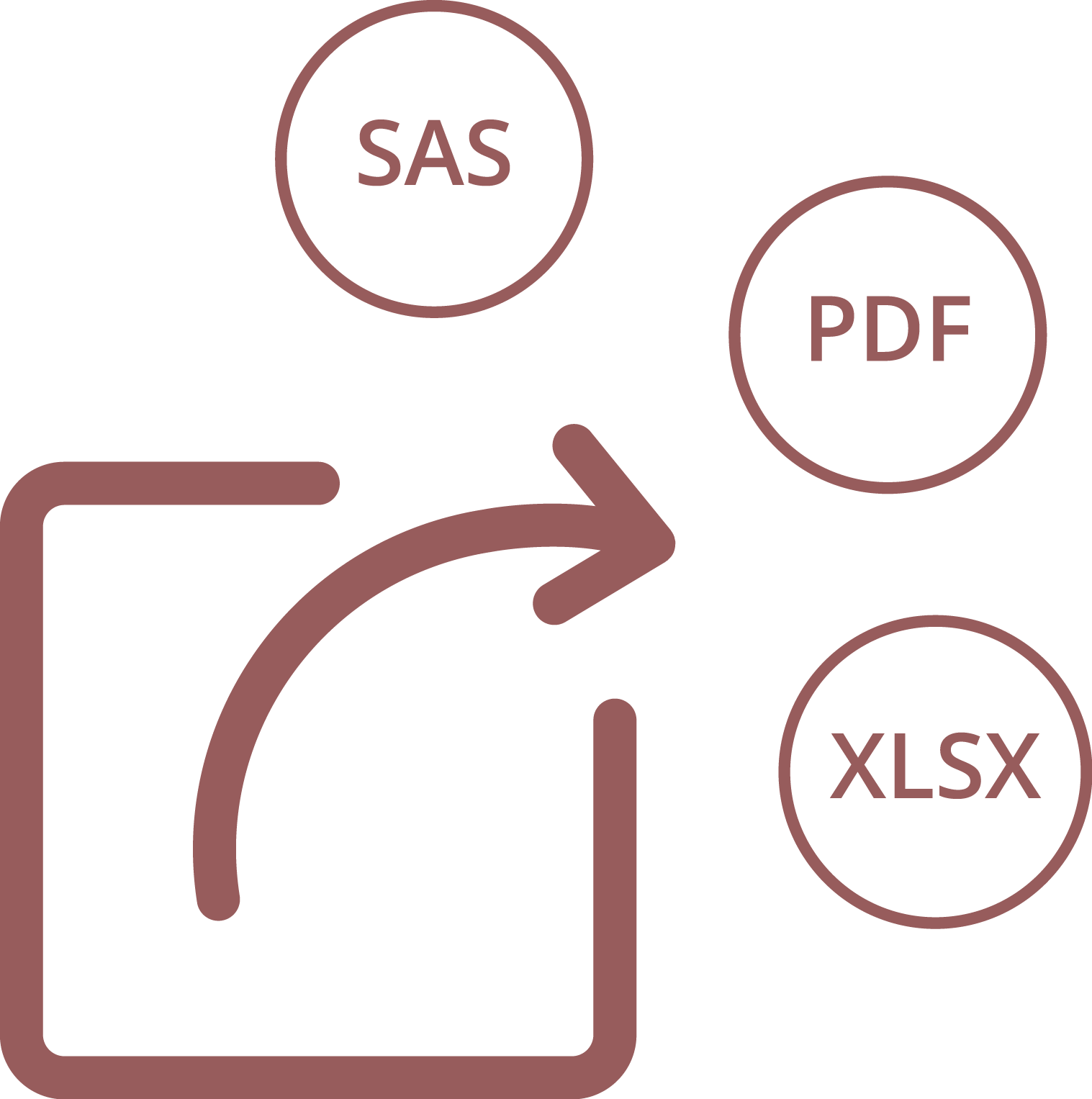
Export
Data can be exported to SAS, Excel and PDF. Several options can be configured, such as the data type, file format, included data or missing values management.
The setup of each export is saved and can be re-used later, to ensure homogeneity in the review and analysis.

Medical coding
ClinInfo's CDMS is compatible with MedDRA, WHO Drug B and WHO Drug C dictionaries, as well as with any kind of specific dictionary.
The coding interface is used for quick coding by grouping terms to code, and automated reports are there to easily detect any discrepancy or abnormality in the coding.
The system also includes an intelligent auto-coder that can code terms based on the dictionary itself as well as choices previously made.
ADVANTAGES



TRY OUR APPS FOR FREE
